FAQs
The FAQs serve as a guide and support for reflection and are subject to change. Each project is assessed in the light of its specificity and context, and only the decision of the cureg is binding.
01. What should I do if I want to change a project that has already been approved by the Committee?
We must be informed of any changes to a project already approved by the Committee (end date, number of participants, procedure, etc.) before the project’s end date. You can get the relevant form by emailing commission-ethique@unige.ch and providing the following information:
- Applicant’s full name
- The project title
- The project number, as indicated on the initial approval form
02. How much does it cost to have a project assessed?
This service is currently free of charge.
03. If I’m involved in a research project that has already been approved by another research ethics committee, do I still need to submit the project to CUREG2.0?
Decisions by other university or public research ethics committees are usually recognized by CUREG2.0. However, we may require you to take additional steps, so you should contact CUREG2.0 before collecting data or carrying out any other procedures.
04. How do I ensure my study’s participants can provide free and informed consent?
4.1 Written consent
As part of the consent process, you may use a pre-study consent form (CF). This should allow participants to make an informed decision about whether they want to participate in the study. In general, a good way to assess your CF is to ask yourself if the person participating in your study might be surprised by aspects of your protocol that they might not expect. If so, then your CF is likely incomplete. Even if the CF informs the person that some things will not be revealed until the end, they should still be warned about what they are about to do.
CFs are typically comprised of the following parts:
- The general purpose of the research activity.
- Required information in order to fulfil the activities (filling out questionnaires, conducting tests, etc.).
- The duration, cost, and potential gain (compensation, gift certificate, etc.).
- Potential risks and how they are controlled by the research protocol.
- The anonymous or non-anonymous nature of the data being collected.
- Whether personal data or sensitive personal data are collected, and if so, which ones.
- Where the data goes (storage and duration of storage, anonymization, etc.), its uses and access rights, deletion, or rectification.
- The individuals who will have access to personal data.
- The researchers’ contact information.
The researchers will determine whether further clarification is necessary. Examples of consent forms and a guide to writing a consent form are available for download by clicking the links below:
CUREG-2023-06-20-GuideRedactionFormulaireConsentment
CUREG-2023-06-20-InformationConsentement_adultes
CUREG-2023-06-20-InformationConsentement_parents-enfants
4.2 Oral consent
In the social sciences and humanities, and particularly in the context of qualitative methods using interviews and/or ethnographic observations, providing written consent can sometimes be problematic or not appropriate. These methods are based on a situated and relational approach with regards to the interaction between the researcher and participant. The expression “investigative relationship” points to a social relationship, and written consent can disrupt this relationship by introducing misunderstandings or preventing the development of mutual trust. This is particularly true when researchers and participants are in regular contact in an ethnographic field (“in situ”).
Furthermore, written consent is deemed not appropriate when participants have characteristics specifically related to literacy or the contractual dimension of the signature, or when the researcher adopts an immersive and/or participatory ethnographic approach.
In cases where written consent is problematic or irrelevant, the applicant to the CUREG must be accompanied by an information and consent protocol in lieu of the written information and consent form. There are two options:
a) mixed written + oral: participants are given a research information sheet (a written document), they are asked questions about the research and their participation, and then are asked to provide oral consent.
b) entirely oral: participants are informed orally, they are invited to ask questions about the research and their participation and are then invited to give their oral consent (e.g., individual oral information in the case of individual interviews; oral information during information sessions at the beginning of the ethnographic fieldwork in a community, etc.).
The information and consent protocol must include the desired option (a or b) and provide details about how it is to be implemented.
05. What preventive measures should be put in place for items related to suicidal ideations?
Including questions on suicidal ideations e.g., item 9 of the Beck Depression Inventory (BDI), is considered potentially problematic because the individual may declare suicidal thoughts or ideas, thus involving the ethical responsibility of the researchers (Green et al., 2015).
If suicidal ideations have been identified, researchers must refer the individual to the appropriate health authorities[1] (for a discussion of this issue see Hom et al., 2017). The solution recommended by the committee is to remove these questions (e.g., use the BDI excluding item 9). If this interferes with the research objectives, an intervention protocol must be created. See the model proposed by Carleton University
Examples are also available in Bailey et al. BMC Medical Ethics, 2020
Response anonymity or confidentiality does not absolve researchers of this responsibility. Where appropriate, a procedure for anonymization must be put in place after problematic cases have been identified. If those responsible for the research choose this option, the consent form should be modified. If the procedure does not allow for the identification of individuals (MTurk, Prolific, etc.), it would be appropriate to automate the receipt or display of an alert to the individual before they exit the application.
If your protocol does not directly address suicide, the solution recommended by the CUREG is to remove questions about suicidal ideations from the questionnaires or to use a measurement tool that does not contain a question related to suicide.
References
Bailey, E., Mühlmann, C., Rice, S. et al. Ethical issues and practical barriers in internet-based suicide prevention research: a review and investigator survey. BMC Med Ethics 21, 37 (2020). DOI: 10.1186/s12910-020-00479-1
Green, K. L., Brown, G. K., Jager-Hyman, S., Cha, J., Steer, R. A., & Beck, A. T. (2015). The predictive validity of the beck depression inventory suicide item. The Journal of clinical psychiatry, 76(12), 1683-1686.
Hom et al., 2017. Ethical issues and practical challenges in suicide research collaboration with institutional review boards. Crisis, 38, 107-114. DOI: 10.1027/0227-5910/a000415
[1] HUG Psychiatry Dept (Urgences secteur psychiatrique)
HUG Young Adults Psychiatry Dept (Unité psychiatrie du jeune adulte)
HUG Young Adult Health (Santé jeunes)
06. I am measuring depression using clinical scales, what are the recommendations?
Including a measure of depression in a protocol always poses an ethical problem (e.g., Clark et al., 2003; https://doi.org/10.1053/apnr.2003.50003). Researchers must therefore ensure that this measure is necessary. Depression scales (e.g., CES-D or BDI) are not diagnostic (Sheehan et al., 2013; https://dx.doi.org/10.1186%2F1472-6939-14-4) and are not designed to measure the intensity of a depressed mood. If the goal is to obtain an estimate of participants’ mood, the committee suggests using tools that specifically measure mood, such as the PANAS.
If the purpose of the research clearly justifies the use of a depression scale, it should not be used alone to suggest to participants a suspected diagnosis of depression. If a participant’s score is above the recommended cut-off, a recommendation can be made to seek advice from their physician. The committee recommends providing participants with a list of accessible centres (thus, depending on their location) and an informative brochure on depression.
The committee would like to draw the attention to the researchers regarding the risk of worrying participants without reason. It is recommended that a high criterion be used before alerting the participant. Certain scales (e.g., the CES-D) must be linked to the presence of other illnesses that may artificially increase the score on this scale.
References:
Clark, P. C., & Dunbar, S. B. (2003). Identifying possible depression in clinical research: Ethical and outcome considerations for the investigator/clinician. Applied Nursing Research, 16(1), 53-59. DOI: 10.1053/apnr.2003.50003
Sheehan, A. M., & McGee, H. (2013). Screening for depression in medical research: ethical challenges and recommendations. BMC medical ethics, 14(1), 1-4. DOI: 10.1186%2F1472-6939-14-4
07. What precautions should I take before using Zoom for a research project?
- UNIGE’s Zoom license must be used.
- The use of Zoom implies the collection of personal data, and you must therefore inform the participants appropriately via the consent form.
- We also invite you to read the document “Good practices in terms of security and confidentiality on Zoom”, available on the university’s website (https://www.unige.ch/enseignement-a-distance/files/4916/3542/4864/11._Zoom11_Securite__confidentialite_v3.1.pdf) and to apply them to your research practices.
- It is not permitted to share the Zoom link of a meeting via social networks.
- If you wish to record interviews via Zoom, a local recording should be used, not a cloud recording.
- Since Zoom does not allow for local audio recordings that are independent of the recording of the image appearing on the camera, it is advisable, if the quality of the recording is sufficient, to use a dictaphone (or any other external recorder) if only an audio recording is planned. Another possibility is to inform the participant that the recording is audio-visual, but that only the audio recordings will be kept and analysed within the research framework (.mp3 files), while the audio-visual recordings will be destroyed immediately (.mp4 files) following local file generation.
- Participants should be asked to change the name that appears on their window before the meeting begins.
- The consent form cannot be sent/collected by Zoom.
In any case, it is necessary to include this additional information in the consent form if testing is to be done virtually. We suggest using the following text:
“If health conditions require it, we will conduct the interviews/tasks/other [to be adapted according to your case] using Zoom software under license with the University of Geneva. The use of Zoom involves the collection of personal data. The Zoom platform is certified by the Swiss-US Privacy Shield agreement. The meeting link will be sent to you by e-mail (your e-mail address will be deleted as soon as we no longer need it) [to be adapted according to your procedure]. Prior to the session, please replace the name on your Zoom display window with a nickname/participant code that we have provided for you [choose the appropriate option for you], so that your identity remains hidden. You will be notified when the audio/video recording begins [if applicable; choose the applicable option for you], and you will be asked to confirm your agreement to the recording verbally. The recording will be stored locally (not on the cloud) and used for strict research purposes. [if only audio recording is needed, specify that audio-visual recordings will be destroyed immediately following the interview].”
08. Virtual reality and risk of cybersickness: what to do?
Ethical question of concern: endangering participants
Immersion in virtual reality can trigger undesirable effects that are well documented in the literature and are referred to as cybersickness. The symptoms are similar to those of motion sickness (kinetosis); those who suffer from motion sickness are more likely to suffer from cybersickness. For this reason, the CUREG recommends that exclusion criteria for those who suffer from kinetosis (self-reported sensitivity to motion sickness) be applied in virtual reality research.
Symptoms of motion sickness are rarely severe and are of short duration. For example, vomiting is rare (less than 15 per 1000).
The symptoms observed systematically in a portion of the population (estimated at 20% in the first 30 minutes) are, in decreasing order of intensity: 1) disorientation (dizziness, imbalance); 2) eye problems (eye fatigue, blurred vision, headaches), 3) nausea (heartache, dizziness).
These risks are reduced in situations where protective factors are present:
– include a negligible delay in the synchronization of the head movements and the movements of the virtual scene (it is recommended to use the most advanced equipment possible, including computers and graphic cards).
– include few rapid movements in the scene (such as scrolling through a landscape).
– age: the literature on the effect of age is sparse, but most existing studies indicate that cybersickness decreases sharply in adulthood, with some authors even indicating that it is non-existent after age 50 (Mousavi et al, 2013).
Nevertheless, even when taking this into account, it must be estimated that between 0 and 20% of participants are likely to experience cybersickness, which justifies the use of a control and support device.
With respect to monitoring, cybersickness can be detected by observing the person’s behaviour (agitation, non-response, etc.), by self-reports, or by a single-item measurement performed regularly (at the end of the habituation phase and then at regular intervals). The task must be interrupted above a predetermined threshold.
In accordance with the literature, the severity of the cybersickness will be moderate at worst. The person should be immediately removed from the helmet and remain seated for at least 30 minutes (average time for symptoms to disappear reported in the literature), and at least until their self-assessment on the single-item measure has dropped to a very low level. In cases of more severe symptoms, the procedures recommended for first aid will be applied.
Reference:
Mousavi, M., Jen, Y. H., & Musa, S. N. B. (2013). A review on cybersickness and usability in virtual environments. In Advanced Engineering Forum (Vol. 10, pp. 34-39). Trans Tech Publications Ltd. DOI: 10.4028/www.scientific.net/AEF.10.34
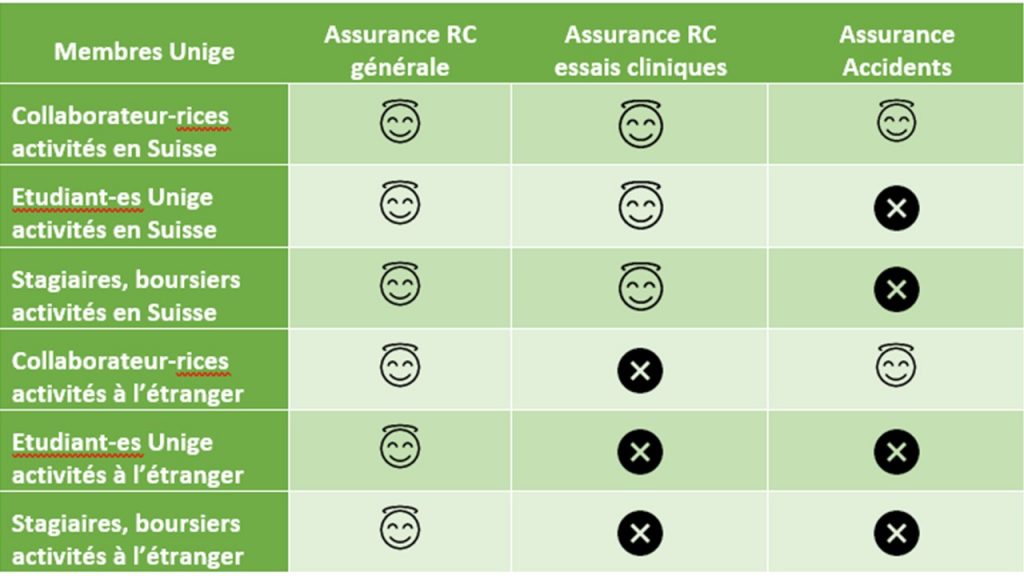
09. Research and Civil Liability Insurance
9.1 What civil liability (CL) insurance coverage(s) is there for Unige researchers?
There are two types of Civil Liability (CL) insurances at the University of Geneva.
- A general CL insurance that covers all “standard” activities of the members of the university, whether they are students, PAT, or PENS collaborators. The activities concerned are those related to the mission of the university, namely teaching, research or services to the community.
- A special CL insurance, called “clinical trials” This is an insurance policy whose benefits are extended to research activities for which risks have been identified by a committee that has assessed the project, such as the CCER or the CUREG. Historically, this CL insurance was put in place for research that included clinical trials, hence its name.
9.2 How are research projects covered by “clinical trials” CL insurance identified?
Research projects for which risks have been identified are reported to the “clinical trials CL” insurance every trimester. Risks that are mainly related to the physical or psychological health of participants and/or researchers are of concern. The activities of all persons involved in the project are covered by the “clinical trials CL” insurance, as long as these activities take place in Switzerland.
9.3 Are students conducting research as part of a bachelor’s, master’s or continuing education project covered by Unige’s general CL insurance?
Yes, students carrying out research work during a bachelor’s, master’s or continuing education program are covered by Unige’s general CL insurance for their research activities when they take place in Switzerland, as is the case for any member of the University of Geneva, provided that they are registered at the university.
9.4 Are students carrying out research involving risks related to the physical or psychological integrity of participants and/or researchers covered by the Unige’s “clinical trials CL” insurance?
Yes, if projects involving students are declared to the “clinical trials CL” insurance by the CUREG following the project’s thorough evaluation procedure.
9.5 How are the activities of those who do not have a contract with Unige covered? We are referring to interns, scholarship holders or doctoral students without remuneration.
A clause stipulates that persons are insured “in the performance of their duties for UNIGE.” The scope of the CL insurance is thus broad. However, an agreement must be formalized between the UNIGE and the scholarship holder or volunteer in connection with the research duties, typically by registration with the university or through an internship contract.
9.6 Does the general CL insurance cover activities carried out abroad?
Yes, the CL insurance is also valid for research activities performed outside Switzerland.
9.7 Does the “clinical trials CL” insurance cover activities carried out abroad?
No, the research activity must take place in Switzerland to be covered.
9.8 Does this CL insurance cover activities carried out in Geneva on participants located abroad?
No, for legal reasons. Research activities carried out by researchers located in Geneva but using participants recruited on European or international platforms will not be covered by the CL insurance. However, if participants come to UNIGE to participate in the research activity, these activities will be covered because Swiss law prevails. Please note that the simple fact of having a questionnaire filled out on foreign territory is not covered by our CL insurance.
10. Research and Accident Insurance (“Assurance Accident”)
10.1 What ACCIDENT INSURANCE coverage is provided for researchers employed by Unige?
Unige employees benefit from the Accident Insurance provided by Unige in line with their contract. This coverage ends upon termination of their employment or service.
10.2 If employees carry out their research abroad, do they still benefit from these two types of insurance, accident, and CL?
Yes, for accident insurance, yes for the “general” CL insurance and no for the “clinical trials” CL insurance. For the latter, it is the “host” institution that provides coverage for the part of the research it conducts, and the coverage must include the students involved.
10.3 What about accident coverage for researchers without a contract but affiliated to Unige (typically master’s students, doctoral or post-doctoral fellows, or volunteers)? What recommendation can be made?
Students should seek personal coverage. The DIFE distributes this information to new students. If they work at least 8 hours per week, their employer will pay for the coverage.
In summary:
11. My research protocol involves home visits to participants. What are CUREG’s recommendations?
For some projects, it is necessary to visit participants’ homes. This can sometimes pose a risk for the researcher. The research supervisor must prioritise the protection of the individuals, especially the students or young researchers, who make the home visit. The CUREG will therefore always carefully examine protocols involving home visits. The CUREG recommends that home visits only be considered if the protocol requires them (and there is no equivalent alternative). In this case, if it does not interfere with the objectives or feasibility of the research, the following aspects may be considered:
- making the visit in pairs.
- the researcher knows the start and end time of the visit.
- those who make the visit are able to send a message to notify the end of the visit.
- If the visit lasts longer than expected, the supervisor must act accordingly.
- those who make the visits are aware of what to do in case of any problem.
12. Preparatory studies for research involving human participants: do I need to apply to CUREG2.0 for certification?
Preparing a research project often requires testing a device on a small number of people, to evaluate its feasibility or to validate part of the material. These tests can also be used to get a feel for the field, to identify potential participants, etc. UNIGE does not require researchers to request a CUREG certificate prior to data collection as part of a preparatory study for a research project. However, this study must be conducted with a small number of participants and is not to be published as the main study in a scientific article. It is explicitly associated with a larger research project. The fact that a certification is not required does not absolve those responsible from respecting the ethical principles of research involving human participants.
13. If I collect personal data as part of my research, what should I look out for?
Preamble:
The university law does not provide a legal basis for the processing of research data and therefore the Law on Public Information, Access to Documents, and Protection of Personal Data (LIPAD) applies to the activities of the university (and therefore to its employees). This cantonal law governs the processing of personal data, sensitive personal data, or the establishment of personality profiles in the context of a research project, except for research projects that fall within the scope of the Federal Act on Research involving Human Beings (HRA) and that are submitted to the Cantonal Research Ethical Committee (CCER).
Consequently, all research projects that do not fall within the scope of the HRA and that involve the collection of non-anonymous data are affected by the LIPAD.
Personal data is defined as any piece of information or group of information that allows for the unequivocal identification of a natural or legal person under private law, such as the identity, date of birth, postal address, e-mail address, etc.
Sensitive personal data and personality profiling are a specific category of personal data that require additional steps. For more details, please refer to the FAQ n° 15.
By processing, we mean (art. 4; letter e – LIPAD): “any operation relating to personal data – regardless of the means and procedures used – in particular the collection, storage, use, modification, communication, archiving or destruction of data.”
Thus, a certain number of principles must be respected, here we list the main ones:
- Principle of proportionality (art. 36 LIPAD): you may only collect personal information that is useful for your purposes: for example, if you need the age but the exact date of birth (01.01.1962) is not useful, then you should not ask for it.
- Purpose principle (art. 35 al. 1 LIPAD): you must tell your participants what information you want to collect about them and the purpose of its collection i.e., why you are collecting this information. This must be done at the time of collection of this information. You may use the data collected ONLY for the purposes originally stated to the participants.
- Principle of security (art. 37 LIPAD): you are obliged to protect this information against any unlawful processing and to ensure its confidentiality by putting in place appropriate protective measures.
- The principle of the right to forget (destruction of data – art. 40 LIPAD): as soon as this information is no longer necessary for your purposes, you must destroy For example, as soon as the name or contact information of a person is no longer useful, this information must be deleted from your files, notes, documents, etc.
Since November 16, 2021, it is no longer necessary to declare files containing personal data to the cantonal Data Protection and Transparency Officer (PPDT). Thanks to the actions taken by UNIGE’s DPO[1] in consultation with UNIGE’s legal affairs department and the CUREG, a generic declaration of files to the catalogue from the cantonal authority is now sufficient for all research projects evaluated by the CUREG and in which personal data are processed. The PPDT has agreed given that the controls relating to the processing of personal data in a research context carried out by the CUREG and the DPO are well supervised.
[1] Alain Jacot-Descombes was appointed as UNIGE’s Data Protection Officer (DPO) in April 2021.
14. What is meant by sensitive personal data or personality profiling within the context of the LIPAD?
According to article 4 (b and c), the law defines these notions as follows
Sensitive personal data: “personal data on religious, philosophical, political or trade union opinions or activities, health, intimate sphere or ethnicity, social assistance measures, criminal or administrative proceedings or sanctions.”
For example, if you ask a mother, what illness her child has, then you are processing (collecting) sensitive personal data. In other words, if you collect the diagnosis(es) (dyslexia, autism, diabetes, etc.) of a person (adult or minor) then you are processing (collecting) sensitive personal data.
Here is another example: if, in the context of a research project, you ask people about their religious practices, you are also processing (collecting) sensitive personal data under the LIPAD.
Consequence: an authorization request to the State Council (Conseil d’Etat) is necessary (see FAQ 15).
Personality profiling: “a collection of data allowing an assessment of the essential characteristics of a natural person.”
This notion remains vague and is not very operational. We sought to clarify this definition with the DPO, who sent us the definition of this concept in the new Federal Act on Data Protection (DPA – Article 5, letters f and g, dated June 7, 2022, this text has not yet been implemented):
- “f) profiling: any form of automated processing of personal data consisting in using such data to evaluate certain personal aspects relating to a natural person, particularly to analyse or predict aspects concerning work performance, economic situation, health, personal preferences, interests, reliability, behaviour, location or moving of the physical person.
- g) high-risk profiling: any profiling that entails a high risk to the personality or fundamental rights of the person of concern, since it leads to matching of data that allows the essential characteristics of the personality of a physical person to be assessed.”
For research, the CUREG considers that a research project deals with personality profiling when tools (quantitative or qualitative) are used that are based on scientific approaches to personality traits, and that allow the respondent to be described on several fundamental dimensions of their personality (e.g., profiling on the “Big Five” dimensions). In this case, the CUREG will invite the person in charge to contact the DPO to check with them whether steps with the State Council (Conseil d’Etat) are necessary (see FAQ 15). Measures of attitudes towards oneself (e.g., self-esteem), values (e.g., solidarity), or single personality traits (e.g., extraversion) are excluded.
15. What steps should I take if I collect sensitive personal data/personality profiles as defined by the LIPAD throughout the course of my research?
Reminder: for data to be considered sensitive personal data/personality profiling, the data must be processed in a non-anonymous way i.e., associated with personal data. For anonymous research (starting from the point of data collection), there is no sensitive personal data nor personality profiling.
The legislator has considered that sensitive personal data/personality profiles are information deserving of a higher level of protection, with the sole aim of protecting the fundamental rights of the individual.
Thus, according to article 41, paragraph 1, letter f of the LIPAD, it is necessary to obtain an authorization from the State Council (Conseil d’Etat) prior to processing sensitive personal data (SPD) or personality profiling. As a reminder, data collection is already considered by the law as a type of processing.
The authorisation request from the State Council must be done as soon as the research project has been accepted by the CUREG.
-
- Procedure:
- From the CUREG’s perspective:
- If a project is concerned by this procedure, the CUREG informs the research supervisor and asks them if they can transmit the revised version to the DPO so that he can verify anything related to the protection of personal data corresponds to the legal rules. He may even ask for modifications regarding solely this issue.
- From the research supervisor’s perspective:
- Based on the model provided by CUREG, the research supervisor drafts a letter addressing the State Council.
- To draft this letter, we invite you to contact Mr. Alain Jacot-Descombes, DPO of the University of Geneva (pdt@unige.ch).
- The person in charge will be responsible for transmitting any useful documents to the State Chancellery.
- The research supervisor will receive a decision from the State Council.
- From the CUREG’s perspective:
- Procedure:
- Special cases:
- If you collect your data anonymously, such as in the following cases:
- Distribution of an online questionnaire, access to anonymous or anonymised databases
- Setting up a procedure that ensures that at no time can you link participants’ responses to their identity
- In these cases, it will not be necessary to apply for authorisation from the State Council for the processing of sensitive personal data/personality profiling within the LIPAD’s framework since you will at no time be aware of the identity of the participants.
- If you collect your data anonymously, such as in the following cases:
16. I am collecting personal data, but my research takes place abroad. Do I still need to follow the LIPAD’s regulations?
The answer is yes.
In this context, it is not the site of the data collection that determines whether LIPAD regulations apply. It is the fact that the data is processed and collected by UNIGE employees that determines whether this law applies. Thus, if you collect sensitive personal data or conduct personality profiling, it will be necessary to take steps with the State Council (Conseil d’Etat) (see FAQ 15).
17. Using online platforms
What is a “cloud” platform?
- An online computer service offered via the internet by a service provider outside UNIGE to carry out certain operations necessary for research. These operations can consist of data collection, storage or analysis, participant recruitment, or online experiments.
What are the security criteria to watch out for when choosing a “cloud” platform for my research?
- The level of sensitivity of the information that will be collected or processed. (Criteria for information classification are available at: https://www.unige.ch/cybersecurite/politiques/classification-de-linformation, only available in French)
- The site of the provider and data host: a Swiss provider and host are recommended. If this is not possible, additional compliance measures will be required in accordance with the document available at: (LINK TO BE CONFIRMED).
- The contractual and technical terms of implementation. (The detailed list of these can be found at: https://www.unige.ch/cybersecurite/pour-les-it-people/regles-minimales-de-securite/solutions-cloud-saas, only available in French)
What are the ethical criteria related to the use of “cloud” platforms?
These criteria can be assessed by asking the following questions about:
- Necessity: is the research unable to be conducted using one of the IT solutions tested and/or offered free of charge by the institution?
- Fair compensation: is the compensation adapted to the standard of living and does not constitute a remuneration? Is a geographical targeting possible in order to adapt the compensation to the local standard of living? Is it customary practice in the given field of research?
- Research transparency: are the purposes and methods of data processing clear? Is consent obtained in the same way as it is for “offline” research?
- Participant supervision: are the necessary guidance and support procedures put in place for sensitive issues?
- Participation at free will: does the researcher guarantee anonymity and confidentiality with respect to local authorities? The possibility to withdraw from the activity at any time and the absence of coercion to participate?
The platforms recommended by the institution for research purposes are the following:
- An institutional version of LimeSurvey has been approved for even highly sensitive data collection, available at: https://catalogue-si.unige.ch/en/limesurvey.
- For storage of your data, the institutional storage system is recommended: https://catalogue-si.unige.ch/en/stockage-recherche.
If I plan to use a platform that is not yet “recommended”, what prior steps should I take?
- We advise you to contact the UNIGE’s data protection officer (DPO) who will be able to evaluate the use of a cloud solution for your research purposes.
- The following form can be sent to the DPO to evaluate the cloud solution you would like to use for research purposes.
- Depending on the sensitivity of the data you plan to process, the provider and host sites, and the technical and contractual terms and conditions, you will need to obtain explicit approval from the Rectorate before using the platform. Based on your request, the DPO will be able to specify the prerequisites required by UNIGE.
18. Usage du terme “race” en recherche : questions éthiques
Le concept de “race” a une longue histoire et a été utilisé dans de nombreux contextes différents. Bien que son usage ait varié d’une époque à l’autre, il a toujours été lié à une volonté de construire et imposer des différences prétendument biologiques et/ou culturelles entre les groupes humains, et son développement dans le contexte scientifique du 19e siècle est indissociable d’une volonté de domination, de contrôle et de pouvoir, notamment en contexte colonial. Dans la seconde partie du 20ème siècle, ce concept a été remis en question à la fois sur le plan scientifique et sur le plan éthique. Les progrès de l’anthropologie et de la génétique ont montré que les différences génétiques entre les populations humaines étaient minimes, que leurs variations phénotypiques étaient continues et conditionnées par les environnements, et que les classifications raciales étaient de ce fait arbitraires et sans fondement biologique rationnel. Par ailleurs, des recherches en sciences humaines et sociales ont montré que les distinctions raciales sont socialement construites sur la base d’identités culturelles et historiques, et que les catégories changent en fonction du contexte. Aujourd’hui, le terme “race” est néanmoins encore utilisé dans le langage commun pour désigner des groupes humains sur la base de critères biologiques et/ou socio-culturels flous, ce qui accentue des stratifications sociales et engendre des discriminations. Dans ce contexte, quelles sont les recommandations éthiques liées à l’usage du concept de « race » dans une recherche scientifique ?
Dans certains pays, comme les USA, le Canada ou la Grande Bretagne, le terme « race » fait partie du langage courant et, même si le concept est contesté (1, 2), il est utilisé dans des cadres légaux (recensement, etc.). Dans d’autres pays, comme la Suisse ou la France, son usage est généralement évité au nom des valeurs d’universalisme et d’égalité (3–5). Ces restrictions d’usage font aussi polémique, dans la mesure où elles sont accusées d’empêcher la mise en évidence de faits sociaux comme la discrimination à l’embauche, ou le contrôle au faciès (6). Les chercheurs et les chercheuses peuvent donc parfois se retrouver en tension entre l’incongruité du concept de race d’un point de vue biologique, et l’évidence de sa réalité dans le monde social (7).
Ainsi, même s’il est admis que la notion de race n’a pas de sens d’un point de vue biologique, elle peut être pertinente dans le cadre de recherches qui visent, par exemple, à examiner comment des différences phénotypiques perceptibles peuvent être associées à une hiérarchisation sociale ou à des rapports interpersonnels particuliers (8).
Cependant, étant donné le sens controversé que véhicule ce mot et son caractère sensible, la CUREG examinera avec attention les justifications proposées par les chercheurs ou les chercheuses qui l’utilisent. À la suite d’une consultation d’expertes et des membres de la commission plénière de la CUREG, des préconisations ont été définies. Les projets seront examinés dans ce cadre.
- Le terme « race » ne peut être utilisé que dans un contexte où son usage est clairement justifié, par exemple lorsqu’il s’agit de mesurer le niveau d’adhésion à des thèses racistes, ou lorsque les acteurs et les actrices concerné-es s’approprient le concept dans des revendications contre toute forme de discrimination raciale.
Mais la plupart du temps, Il est conseillé de ne pas utiliser ce terme. Cela vaut pour le matériel et pour les textes associés (titre de la recherche, résumé, description du protocole, etc.).
- Il est également recommandé de ne pas utiliser de dénominations ou de catégorisations suggérant l’existence d’une typologie raciale ou renvoyant une vision réductrice de la diversité biologique et/ou culturelle humaine, qui posent les mêmes problèmes éthiques que le mot « race » ; par exemple « caucasien », « noir », « phénotype asiatique », etc.
- L’usage de termes ou expressions comme « ethnie », « appartenance ethnique », « origine », « ethnie, ethnicité et autres circonstances particulières », « origine ethnique », « origine ethnique, sociale et territoriale », « ancestralité », etc., est souvent proposé comme alternatives éthiquement correctes. Mais ces appellations ambiguës ou mal définies doivent être utilisées avec précaution et toujours en justifiant leur usage dans le cadre de la demande d’évaluation éthique.
- Lorsqu’une variable liée à l’apparence physique des participant-es est mesurée, les chercheurs et les chercheuses doivent justifier la nécessité scientifique du recueil de cette donnée. Il est recommandé de mettre la priorité sur des termes spécifiques plutôt que collectifs, et de choisir des descripteurs de même nature pour tous les individus. Par exemple, pour étudier l’effet de la « race » sur le contrôle au faciès, il est préférable de recueillir une donnée comme la couleur de peau, si possible à l’aide d’une mesure qui laisse libre cours à toute la diversité de cette caractéristique (par exemple, les palettes continues de teintes de peau existant dans le commerce).
- La mesure de l’auto-identification d’une personne à une « race » doit être évitée et, si nécessaire, remplacée par des critères d’auto-identification choisis avec beaucoup de précautions. Il n’est pas rare de lire des questions comme « quelle est votre race/ethnicité : Caucasien, etc. ». Une formulation neutre (ex : « je me décrirais comme… ») proposant une réponse ouverte est recommandée. Si des réponses fermées à choix multiples doivent être soumises, l’option « aucune des réponses ne me décrit » doit être obligatoirement présente.
Références
- J. Nicol, B. Osazuwa, Les mots pour parler de race et d’ethnicité : une terminologie en évolution. Notes de la Colline, (available at https://notesdelacolline.ca/2022/01/31/les-mots-pour-parler-de-race-et-dethnicite-une-terminologie-en-evolution/).
- Committee on the Use of Race, Ethnicity, and Ancestry as Population Descriptors in Genomics Research, Board on Health Sciences Policy, Committee on Population, Health and Medicine Division, Division of Behavioral and Social Sciences and Education, National Academies of Sciences, Engineering, and Medicine, Using Population Descriptors in Genetics and Genomics Research: A New Framework for an Evolving Field (National Academies Press, Washington, D.C., 2023; https://www.nap.edu/catalog/26902).
- P. Mahon, La notion de « race » dans le droit suisse : à jeter aux oubliettes ? TANGRAM 44 (2020), (available at https://www.ekr.admin.ch/f585.html).
- M. Merenda, La définition de la race par les autorités judiciaires suisses. TANGRAM 44 (2020), (available at https://www.ekr.admin.ch/publications/f587.html).
- C. Gombault, G. Grenet, L. Segurel, L. Duret, F. Gueyffier, P. Cathébras, D. Pontier, S. Mainbourg, A. Sanchez‐Mazas, J. Lega, Population designations in biomedical research: Limitations and perspectives. HLA. 101, 3–15 (2023).
- R. Gremaud, Les discriminations raciales ne sont pas chiffrées en Suisse. ma RTS (2020), (available at https://www.rts.ch/info/suisse/11397852-les-discriminations-raciales-ne-sont-pas-chiffrees-en-suisse.html).
- J. L. Martin, K.-T. Yeung, The Use of the Conceptual Category of Race in American Sociology, 1937–99. Sociological Forum. 18, 521–543 (2003).
- C. L. Ford, N. T. Harawa, A new conceptualization of ethnicity for social epidemiologic and health equity research. Social Science & Medicine. 71, 251–258 (2010).
19. Ma méthode de recherche ne me permet pas ou difficilement de recueillir un consentement. Comment faire ?
La CUREG a identifié deux cas pour lesquels il peut y avoir une demande de dérogation au consentement.
CAS 1 : observation sans manipulation de l’environnement.
Il s’agit des études où les chercheurs/euses observent les comportements spontanés, sans aucune intervention ni manipulation. Par exemple il peut s’agir d’une recherche où on observe la vitesse des conducteurs/trices sur une portion de route en fonction du modèle de voiture. Ou bien une étude où l’on compte le nombre de prises de parole dans un cours en fonction du sexe des élèves. Dans ce cas, la CUREG peut examiner une demande argumentée d’absence de consentement, aux conditions suivantes :
– L’étude est anonyme dès le recueil des données (pas de voix, vidéo, photo, etc.).
– L’observation se fait en cadre naturel (situation quotidienne, publique, espace public environnement usuel) ou lieu public (p. ex. stade, bibliothèque, musée, planétarium, plage ou parc) ou dans un environnement numérique (p. ex. groupes en ligne) ou dans des espaces privés ou protégés (p. ex. clubs privés, clubs sportifs ou organisations).
– Le chercheur ou la chercheuse n’est pas intervenu-e sur la situation.
– Il n’y a pas de conflit d’intérêts.
CAS 2 : observation avec manipulation de l’environnement à l’insu des personnes.
Il s’agit d’observations en situation naturelle, mais le chercheur ou la chercheuse induit une manipulation de l’environnement afin d’en tester les effets. Par exemple, une étude pourrait tester différentes formes de courriers d’invitation au don du sang, afin de voir laquelle est la plus efficace. Ou bien, des spécialistes en comportements environnementaux pourraient tester différents messages pour inciter les usagers d’un bâtiment à utiliser les escaliers plutôt que les ascenseurs. Dans ce cas, la CUREG peut examiner une demande argumentée d’absence de consentement, aux conditions suivantes :
– L’étude est anonyme dès le recueil des données (pas de voix, vidéo, photo, etc.).
– L’observation se fait en cadre naturel (situation quotidienne, publique, espace public environnement usuel) ou dans un lieu public (p. ex. stade, bibliothèque, musée, planétarium, plage ou parc) ou dans un environnement numérique (p. ex. groupes en ligne) ou dans des espaces privés ou protégés (p. ex. clubs privés, clubs sportifs ou organisations).
– Il n’y a pas de risques induits par la manipulation.
– Il n’y a aucune forme de consentement possible, a priori ou a posteriori, sans mettre en péril la recherche.
– La prise de risques éthique est justifiée par les enjeux de la recherche.
– Il n’y a pas de conflit d’intérêts.
Dans tous les cas, lorsque cela est possible, les chercheurs et les chercheuses sont invité-es à prendre des mesures pour informer le public concerné qu’une recherche est en cours et de diffuser le nom et les coordonnées de la personne responsable (par voie d’affichage ou de documents distribués lors de l’achat des tickets d’entrée par exemple).
20. Pourquoi la CUREG me demande de m’adresser à la Commission Cantonale d’Éthique de la Recherche ?
Les règles fédérales Suisses, reposant sur la loi sur la Recherche sur l’Humain (LRH) et son application, imposent aux chercheurs et aux chercheuses de passer par la CCER dans les cas suivants :
- recherche relative aux causes et aux fondements des maladies, y compris les aspects psychosociaux[1].
- les études portant sur la mesure de l’activité cérébrale (IRM, EEG, etc.) ou de la physiologie périphérique (ECG, mesure de l’activité électrodermale, etc.) durant une observation expérimentale du comportement (scénario Y de la prise de position swissethics[2]).
- les essais cliniques (interventions)[3].
Parfois, même si votre recherche tombe dans l’une de ces situations, la CCER considère qu’elle ne souhaite pas entrer en matière. Dans ce cas, la CUREG accepte de traiter votre dossier. Mais vous devez fournir une attestation de non entrée en matière de la part de la CCER. Pour des raisons juridiques, cette attestation doit être fournie pour chaque nouveau projet soumis.
[1] Message sur la loi fédérale relative à la recherche sur l’être humain du 21 octobre 2009, section 1.8.1.3 p. 7294.
[2] Guide de swissethics : demandes dans le domaine des neurosciences du 14 janvier 2016
[3] Ordonnance sur les essais cliniques hors essais cliniques de dispositifs médicaux du 20 septembre 2013
21. Recherche à visée de formation : dois-je soumettre à la CUREG ?
Certaines situations impliquent un recueil de données auprès de participant-es humain-es, mais avec un objectif de formation uniquement (par exemple, maîtriser une technique de recueil de donnée ou avoir une expérience du processus de recherche) et sans visées scientifiques. Cela peut être le cas de travaux pratiques ou de travaux de recherches en Bachelor. Dans ces cas une attestation éthique n’est pas nécessaire et la CUREG n’entre pas en matière. Les aspects éthiques sont sous la responsabilité des enseignant-es. Si ces travaux s’adossent à un projet de recherche plus large, celui-ci doit être traité dans le cadre de la procédure de dépôt de projets.
22. J’utilise des applications, des logiciels ou des plateformes externes à l’UNIGE pour recueillir mes données (Qualtrics, RealLife Exp, Gorilla, Prolific, Field Notes, etc.), quelles précautions dois-je prendre ?
Le recours à des outils externes à l’UNIGE pour recueillir des données de recherche est devenu très courant. Cette pratique pose trois problèmes potentiels : 1) la conformité légale du stockage des données 2) l’usage potentiel des données par le fournisseur de l’outil et 3) le consentement éclairé des participant-es.
- Conformité légale. Compte-tenu de la rapidité du développement des produits, les services internes de l’UNIGE ne peuvent pas valider l’usage de tous les outils offerts aux chercheurs et aux chercheuses. Il relève donc de votre responsabilité de vérifier la compatibilité de celui que vous utilisez avec les lois en vigueur en Suisse (notamment la LIPAD). Cette information se trouve généralement de façon déclarative dans les politiques de confidentialité de votre fournisseur.
- Usage des données par le fournisseur. Une conformité légale peut être compatible avec l’utilisation des données recueillies par le fournisseur à des fins commerciales. Cela peut aller de la simple utilisation pour améliorer l’outil, à la commercialisation des données fournies par vos participant-es, par exemple pour entraîner des intelligences artificielles. Il vous appartient de vérifier les pratiques de votre fournisseur.
- Consentement des participant-es. Si le fournisseur peut utiliser les données des participant-es (même de façon anonyme et agrégée) à d’autres fins que celles de votre recherche, les participant-es doivent en être informé-es à l’aide du formulaire de consentement. Une phrase type pourrait être « Nous utilisons une application tierce pour recueillir vos réponses. Notre fournisseur peut utiliser ces données [le cas échéant préciser : de façon anonyme et agrégée] à des fins privées [éventuellement préciser lesquelles]. »
23. I conduct research within an organization: what is the difference between the permission obtained from the organization’s authorities and the consent of the participants?
Distinction between Entry Agreement and Participant Consent
When conducting research with human participants within an organization (business, institution, association, school, etc.), it is often necessary to obtain the agreement or authorization from a person with authority in that organization (a person holding the status of “gatekeeper”).
The agreement to conduct research within an organization does not imply the consent of the members of that organization to participate in the research. The term “consent” implies the explicit agreement of the participants in the study; an organization cannot give consent on behalf of the participants. The Swiss Academy of Sciences’ Code of Scientific Integrity defines it as follows:
“Informed consent is a procedure by which a researcher obtains and maintains permission from a person, or their legal representative, for that person to participate in a research study” (ASS, 2020, p. 13).
Who are the Participants? Who Consents?
The participants, in other words, the individuals from whom consent should be obtained, are the people from whom data is collected.
In research conducted within an organization, the agreement is therefore not sufficient, and obtaining the consent of each participant is necessary (except in special cases, see FAQ-19).
A Suggestion for Further Reading:
Canadian Code “Ethics of Research Involving Humans”, Tri-Council Policy Statement (TCPS 2), 2022, https://ethics.gc.ca/fra/policy-politique_tcps2-eptc2_2022.html
24. Puis-je ne pas anonymiser les données de ma recherche dans les communications des résultats ?
Dans le cas où la recherche est compatible avec une non anonymisation des données, le ou la chercheuse peut décider d’inclure l’identité des participant-es dans la communication des travaux, à condition d’en informer explicitement les participant-es et d’obtenir leur accord écrit. Cela est notamment le cas dans les études en sciences sociales mais également dans des travaux en traductologie, où le recours systématique à l’anonymisation n’est pas réalisable, pas souhaité ou pas souhaitable. Il est nécessaire d’attirer l’attention des participant-es sur le fait que les données publiées ne peuvent pas être modifiées.
25. Comment communiquer les résultats de la recherche aux participant-es ?
Les participant-es à la recherche doivent être informé-es, dans le cadre de la procédure de consentement, de la manière dont ils/elles recevront un retour des résultats.
Donner un retour des résultats aux participant-es d’une recherche est une démarche essentielle pour reconnaître et valoriser leur contribution. Les personnes impliquées — qu’elles soient interviewées, observées ou qu’elles répondent à un questionnaire — partagent volontairement leur temps et leurs savoirs, motivées par des idées, des attentes ou une curiosité légitime quant aux conclusions de l’étude. Il est ainsi recommandé d’adresser des remerciements aux participant-es, généralement de manière anonyme, dans chaque publication reposant sur leur contribution. En leur permettant d’accéder aux résultats, les chercheur-es favorisent une relation fondée sur la transparence et le respect mutuel. À défaut de cette démarche, la recherche risque de basculer dans une logique d’ « extractivisme », où les participant-es seraient réduit-es à de simples sources de données, sans reconnaissance de leurs apports ni retour sur les résultats finaux.
Nous préconisons d’adopter, dans la mesure du possible, une position proactive qui permette aux participant-es de prendre connaissance des résultats de manière autonome, sans être obligé-es d’en faire une demande explicite. Alternativement, vous pouvez fournir une adresse email sur laquelle les participant-es peuvent vous contacter pour obtenir les résultats de la recherche.
Différents formats de restitution sont possibles, entre autres des « lay-summary », des vidéos explicatives courtes, la publication sur des blogs ou réseaux sociaux scientifiques des principaux résultats, ou encore des brèves présentations orales. Le format choisi et l’organisation du retour des résultats aux participant-es dépendent du contexte de la recherche. Ils peuvent varier selon le degré d’anonymisation, le nombre de participant-es, leur âge, leur profil ainsi que le niveau de formation ou leurs compétences linguistiques.
En cas de non-anonymisation, il est par exemple possible d’envoyer un résumé des résultats aux participant-es de la recherche. Les participant-es peuvent également, s’ils ou elles le souhaitent, fournir une adresse e-mail à laquelle les résultats leur seront envoyés. Dans ce cas, il est impératif de mentionner clairement dans le formulaire de consentement que cette adresse e-mail sera conservée uniquement à cette fin et supprimée une fois les résultats communiqués. En cas d’anonymisation, il est possible d’indiquer un site web ou les résultats seront mis à disposition sous forme accessible, en précisant une date approximative de publication.
Enfin, dans le cadre de recherches menées en collaboration avec des associations ou des institutions, il est possible de partager les résultats via ces partenaires. Ceux-ci peuvent jouer un rôle de relais, notamment en organisant des événements de restitution spécialement conçus pour les participant-es. Ces rencontres offrent un espace d’échange et permettent de rendre les résultats accessibles de manière interactive et conviviale. Alternativement, les participant-es peuvent être invité-es à contacter directement l’association ou l’institution partenaire pour obtenir les résultats.
26. Je souhaite collecter le sexe ou le genre de mes participant-es, ou bien poser d’autres questions liées au sexe ou au genre. Quelles sont les recommandations de la CUREG ?
Demander à une personne quel est son sexe ou son genre n’est pas une question anodine. Cette interrogation soulève de nombreux enjeux éthiques, sociaux et identitaires. En effet, les notions de sexe et de genre ont donné lieu à des élaborations scientifiques qui ne recoupent pas systématiquement les représentations communes des sexes. Ce décalage entre terminologie scientifique, réalité vécue et attentes sociales crée des tensions. Par exemple, certaines personnes réagissent négativement lorsque des approches non binaires du sexe sont proposées, les percevant comme une remise en cause de repères traditionnels ou comme une forme d’idéologie. À l’inverse, des personnes se sentent invisibilisées, voire blessées, lorsqu’on leur impose une catégorisation binaire, fondée principalement sur des caractéristiques biologiques.
Dans ce contexte, les chercheurs et les chercheuses, notamment lorsqu’ils ou elles ne sont pas spécialistes de ces sujets, peuvent rencontrer des difficultés à formuler des questions liées au sexe ou au genre. Notre objectif est d’identifier des points d’attention et de fournir des pistes de formulations qui soient à la fois scientifiquement rigoureuses, adaptées aux questions de recherche, et qui minimisent les tensions potentielles qui pourraient se présenter. Notons que les risques éthiques ne se limitent pas aux participant-es. Par exemple, les chercheurs et les chercheuses peuvent être négativement impacté-es (controverse virale, tempête médiatique, etc.).
C’est dans ce cadre global que la CUREG a décidé d’engager une consultation sur les enjeux éthiques liés à la manière de collecter les informations relatives aux concepts de sexe ou de genre. L’objectif était de définir un certain nombre de recommandations aux chercheurs et aux chercheuses.
La consultation s’est faite auprès :
- d’un groupe de travail composé d’expert-es du domaine du sexe et du genre et de chercheurs et chercheuses de l’Université de Genève, ainsi que d’une représentation des groupes socialement ou politiquement impliqués dans la question du genre à Genève
- du département de l’instruction publique de l’état de Genève et du service de recherches dans les écoles
- du dicastère Vivre-ensemble (commission égalité de l’UNIGE)
- de l’assemblée plénière de la CUREG
Définitions
La distinction entre sexe et genre n’est pas systématique et il n’est pas rare de les voir utilisés de façon interchangeable dans le langage courant et administratif. Néanmoins, les recherches scientifiques ont montré que ces deux concepts sont distincts et nécessaires pour comprendre certains phénomènes sociaux qui ne s’expliquent pas par une conception purement biologique du sexe. Des définitions du sexe, du genre et concepts apparentés sont à disposition des personnes souhaitant se documenter sur le sujet. L’objectif de cette FAQ étant surtout d’aider les chercheurs et les chercheuses à travailler avec ces concepts sur le terrain, la CUREG a choisi de ne pas ajouter de définitions mais de renvoyer aux définitions existantes. Il est attendu que les principales définitions soient maîtrisées avant d’entreprendre tout travail de recherche. Le groupe de travail recommande de se référer aux travaux menés de façon indépendante par des expert-es du sujet, notamment (liste non-exhaustive) :
- La prise de position de la commission nationale d’éthique dans le domaine de la médecine humaine. Bien que ce texte traite principalement de la prise en considération de la diversité des identités de genre lors de l’enregistrement officiel du sexe, le document fournit un certain nombre de définitions. Ressource disponible ici :
- Sex and Gender Equity in Research: rationale for the SAGER guidelines and recommended use : cet article en open access ne porte pas principalement sur les concepts de sexe et de genre mais en propose des définitions. Ressource disponible ici :
https://link.springer.com/content/pdf/10.1186/s41073-016-0007-6.pdf
- Examen approfondi de la mesure de l’identité de genre. Il s’agit d’un rapport de la Commission économique pour l’Europe (Conférence des statisticiens européens) qui expose les pratiques et les enjeux de la mesure de l’identité de genre. Une section recense des définitions. Ressource disponible ici :
- La gender toolbox: un travail mené par l’unité « Santé et genre » de Unisanté. Cet outil propose des indicateurs du sexe, et de nombreux concepts qui leur sont apparentés. Ressource disponible ici :
Le tableau 1 recense les définitions des concepts de sexe et d’identité de genre proposées dans les documents cités ci-dessus. Généralement, le terme « sexe » renvoie à la présence de plusieurs éléments biologiques (y compris génétiques ou anatomiques), d’où l’usage fréquent de l’appellation « sexe biologique ». En raison de sa difficulté à être mesuré directement, le sexe biologique est souvent approximé par le sexe assigné à la naissance, lui-même basé sur l’observation des organes génitaux externes chez le nouveau-né ou la nouvelle-née. Le sexe assigné à la naissance n’est pas un indicateur parfaitement fiable du sexe biologique, et la marge d’erreur potentielle doit être prise en considération.
La notion de genre renvoie à des éléments construits sur la base des différences de sexe, et liés à des déterminants sociaux et culturels. Dans le cadre d’un recueil du genre chez des participant-es, les chercheurs et les chercheuses peuvent s’intéresser à différentes dimensions du genre (identité de genre, expressions de genre, etc.). En l’état actuel de la science, il serait sans doute difficile de donner une liste consensuelle des catégories de genre. Il est néanmoins admis que la binarité H/F ne reflète pas correctement la diversité des genres dans la population.
Tableau 1 : principales définitions des notions de sexe et de genre, et indicateurs suggérés dans la gender toolbox
| Prise de position de la CNE | Sex and Gender Equity in Research: rationale for the SAGER guidelines and recommended use | Examen approfondi de la mesure de l’identité de genre | Indicateurs proposés dans la gender toolbox | |
| identité de genre (le genre, lorsqu’il est recueilli au niveau individuel) | « Le concept d’identité de genre fait référence à l’expérience intime et personnelle de son genre. » (p.8) | « Le genre fait référence aux rôles, comportements et identités socialement construits des personnes de sexe féminin, masculin et des personnes de genre divers. » Notre trad. p.1) | « Dans les ouvrages spécialisés, l’« identité de genre» (gender identity) est généralement définie comme le sentiment intime d’être une femme, un homme, ou pas tout à fait l’un ou l’autre, indépendamment du sexe assigné à la naissance. »(p.3) | Femme, Homme,
Femme transgenre, Homme transgenre, Autre (agenre, genre fluide, non-binaire, en questionnement/ pas sûr(e), etc.) – Si « Autre », merci de spécifier (champs libre) |
| Sexe | « Le sexe biologique est déterminé sur la base de caractéristiques telles que les chromosomes sexuels, les organes sexuels primaires, les hormones sexuelles et les autres caractéristiques sexuelles qui en résultent ». (p.9) | « Le sexe fait référence à un ensemble de caractéristiques biologiques chez les humains et les animaux, associées à des traits physiques et physiologiques, notamment les chromosomes, l’expression génétique, le fonctionnement hormonal et l’anatomie reproductive/sexuelle.» (Notre trad. p.1) | N/A | – Sexe assigné à la naissance* : À quel sexe vous a-t-on assigné à la naissance, c’est-à-dire ce qui figure sur votre acte de naissance ? – Femme, Homme
– Caractéristiques sexuelles : idéalement à travers des mesures biomédicales des caractéristiques pertinentes liées au sexe. (Notre trad.) |
* « Sexe inscrit sur votre acte de naissance » peut être une option moins sensible
** Dans sa prise de position, la commission nationale d’éthique préfère « divers » comme troisième possibilité. p.33
Recommandations
Les chercheurs et les chercheuses souhaitant travailler avec les concepts de sexe et de genre sur le terrain sont invité-es à prêter attention aux recommandations suivantes.
Recommandation 1
Si une question relative au sexe ou au genre est formulée, sa présence doit toujours être justifiée. Les raisons peuvent être d’ordre scientifique, liées à des normes de publications, ou à de potentiels risques de pertes d’informations pour les méta-analyses (par exemple ne pas disposer de cette information pourrait créer le risque de manquer de données pour observer des effets de discrimination), etc. Dans tous les cas, les chercheurs et les chercheuses sont invité-es à interroger leur pratique et celle de leur communauté, afin que la présence de ces questions repose sur des arguments forts et non pas sur une routine.
Recommandation 2
Les concepts liés au sexe ou au genre doivent être définis et utilisés à l’échelle où ils sont le plus pertinents pour comprendre, expliquer ou intervenir de manière rigoureuse sur le plan scientifique. En effet, il existe toute une terminologie associée à l’étude du sexe ou du genre, à commencer par les concepts de sexe et de genre eux-mêmes, mais aussi tous ceux qui leur sont liés, par exemple identité de genre, expression de genre, etc. Les chercheurs et les chercheuses doivent pouvoir identifier et définir clairement les informations qu’ils ou elles ont besoin de collecter.
Recommandation 3
La ou les questions posées pour collecter les concepts liés au sexe ou au genre doivent correspondre à la définition retenue dans la recherche (recommandation 2). Par exemple, du fait de la nature multidimensionnelle du concept de genre, poser la question « quel est votre genre ?» devrait être considéré comme trop imprécis pour recueillir l’identité de genre (cf. l’exemple de définition d’identité de genre dans le tableau 1). Les formulations mélangées telles que « Quel est votre sexe/genre » sont à éviter. La formulation « Sexe assigné à la naissance », bien que jugée comme scientifiquement la plus objective, génère régulièrement des réactions négatives. La formulation « Sexe inscrit sur votre acte de naissance » peut représenter une alternative acceptable, si elle est adaptée à la population interrogée (cf. tableau 1).
Recommandation 4
Le fait qu’un concept soit validé par la science n’autorise pas les chercheurs et les chercheuses à l’utiliser sans précautions auprès des participant-es à leurs études. Les chercheurs et les chercheuses doivent respecter les valeurs ou opinions de leur participant-es. Ils ou elles sont donc invité-es à anticiper ces réactions négatives, d’une part en tenant compte des sensibilités de leurs participant-es, et, d’autre part, en fournissant des explications sur leur approche du sexe ou du genre. Dans l’idéal, s’il y a potentiellement un risque auprès de la population cible de l’étude, celui-ci devrait être évoqué et justifié dans le formulaire de consentement.
La collecte du sexe ou du genre peut être particulièrement problématique auprès de certaines populations pour des raisons de vulnérabilité. Il s’agit principalement des populations discriminées en raison de leur genre, et des enfants. En ce qui concerne les populations discriminées, il est recommandé de consulter des membres de la population ciblée afin de discuter du matériel de l’étude dès sa construction. En ce qui concerne les enfants, certains parents expriment leur inquiétude au sujet des effets que certaines formulations pourraient avoir sur le développement psychologique et social de leurs enfants. Il n’y a pas de consensus scientifique sur ce point. Les concepts liés au sexe ou au genre peuvent être recueillis chez les enfants, mais en prenant en compte les différents stades de développement et de préférence en questionnant les parents ou en utilisant des observations indirectes, tout en évaluant le risque de mégenrer l’enfant. Par exemple le sexe enregistré à l’état civil peut être approximé à partir de l’observation de l’expression de genre de l’enfant, si la marge d’erreur est acceptable. Si une formulation potentiellement problématique doit être utilisée auprès d’enfants, le gain scientifique doit être important. Dans ce cas, l’accord parental est normalement requis.
Ressources.
- En cas de doutes sur la manière d’interroger les participant-es, contactez la CUREG : ethique@unige-ch
- Un certain nombre d’associations ou d’institutions suggèrent des méthodes pour recueillir le sexe ou le genre en plusieurs étapes :
The williams institute : https://williamsinstitute.law.ucla.edu/quick-facts/survey-measures/
Nations unies – Conseil économique pour l’Europe : https://unece.org/sites/default/files/datastore/fileadmin/DAM/stats/documents/ece/ces/2019/ECE_CES_2019_19-G1910227F.pdf
- Des propositions de mesures continues du sexe et du genre existent. Par exemple :
https://www.samkillermann.com/work/genderbread-person/
27. Mes données sont-elles réellement anonymes ? Comment gérer le risque de réidentification.
Qu’est-ce que la réidentification ?
La réidentification survient quand une personne peut être retrouvée dans une source de données prétendument anonymes en reliant des combinaisons de quasi-identifiants (p. ex. sexe, code postal, date de naissance, niveau d’études) à d’autres sources de données non-anonymes.
Exemple
Une équipe de recherche a recueilli des données sur la santé mentale des étudiant-es en deuxième année du bachelor en psychologie. Le questionnaire était anonyme. L’âge (en années) et le sexe (M/F/autre) étaient demandés. Par ailleurs, l’administration détient un fichier étudiant-es non-anonyme qui contient aussi l’âge et le sexe. Dans ce fichier la combinaison « Homme/49 ans/B2 psychologie » n’apparaît qu’une fois. Potentiellement, il est donc possible de réidentifier le participant qui correspond à la ligne « homme de 49 ans » dans le premier fichier de données prétendument anonymes.
Quels sont les risques liés à la réidentification pour les chercheurs et les chercheuses ?
La LIPAD donne la définition des données personnelles : « […] toutes les informations se rapportant à une personne physique ou morale de droit privé, identifiée ou identifiable » (souligné par nous). Un fichier avec possibilité de réidentification tombe donc dans le champ de la LIPAD, et pose un risque légal pour les chercheurs et les chercheuses si le cadre de la LIPAD n’a pas été respecté.
Quels sont les risques liés à la réidentification pour les participant-es ?
Le risque de réidentification induit pour les participant-es une menace de divulgation de données personnelles sensibles.
Quelles sont les bonnes pratiques ?
Les chercheurs et les chercheuses sont invité-es à évaluer le risque de réidentification ainsi que les conséquences potentielles.
Concernant le risque, un calcul de k-anonymity (cf. ci-dessous) peut être réalisé. Un seuil de k>4 (au moins 5 personnes correspondent à n’importe quelle combinaison de quasi-identifiants dans le fichier prétendument anonyme) pourrait être considéré comme acceptable. Le risque doit être pondéré par la difficulté de la réidentification. Dans l’ORH (art.25) figure la notion « d’efforts disproportionnés » à mettre en œuvre pour réidentifier la personne. Ainsi si les sources qui permettent la réidentification n’existent pas, ne sont pas accessibles, ou alors moyennant des efforts disproportionnés, le risque peut être considéré comme acceptable. Les chercheurs et les chercheuses doivent porter attention au fait que les nouvelles technologies et les fuites de données peuvent rendre la réidentification plus facile qu’auparavant, notamment lorsqu’un fichier contient beaucoup de quasi-identifiants.
Concernant les conséquences, elles doivent être évaluées sur la base du type de données qui risquent d’être divulguées. Les données anodines (par exemple des temps de réactions sur une tâche de détection d’un signal) peuvent minimiser les conséquences d’une réidentification. Toutes les autres données, notamment celles considérées comme sensibles par la LPD et la LIPAD, celles qui pourraient mettre en danger les participant-es, ou celles qui pourraient être utilisées à des fins commerciales, doivent donner lieu à un traitement attentif du risque de réidentification.
Comment calculer la k-anonymity de votre source de données ?
Considérons que votre source est un fichier anonyme contenant les données de 2000 participant-es âgé-es de 20 à 60 ans. Parmi les données que vous possédez, 3 pourraient être utilisées comme quasi-identifiants : l’âge (en années, 41 modalités), le sexe (H/F/Autre, 3 modalités) et le canton d’habitation (Jura, Neuchâtel, Vaud, Genève, Berne, Fribourg et Valais, 7 modalités). Il existe 41*3*7 = 861 combinaisons des modalités des 3 quasi-identifiants. En comptant les occurrences de chacune de ces combinaisons dans votre fichier, vous constatez que certaines apparaissent rarement. Celle qui apparaît le plus rarement est F/42 ans/Jura. Cette combinaison apparaît 2 fois. Le k est de votre source de données est donc de 2.
Cet indice est très conservateur, car même si l’occurrence n’apparaissait qu’une seule fois dans votre fichier, il faudrait qu’elle apparaisse aussi une seule fois dans la deuxième source non-anonyme pour permettre une réidentification. Dans l’idéal, la k-anonymity devrait donc être calculée sur la seconde source non-anonyme mais cela est rarement possible. Calculée sur le fichier source prétendument anonyme, la k-anonymity reste un bon indicateur du risque attendu dans le pire des scénarios.
Quelles sont les solutions ?
La CUREG recommande d’examiner les possibilités suivantes :
- Augmenter la k-anonymity. Cela peut se faire en continuant à inclure des participant-es jusqu’à atteindre le seuil de k>4, ou bien en excluant les participant-es dont le profil de quasi-identifiants apparaît moins de 5 fois dans la source de données prétendument anonymes.
- Ne recueillir des quasi-identifiants que si cela est strictement nécessaire pour la recherche.
- Recueillir des quasi-identifiants plus difficiles à utiliser pour la réidentification (p.ex. âge en catégories plutôt qu’en années, opinion politique sur un continuum plutôt qu’en catégories).
- Restreindre fortement l’accessibilité du fichier de données prétendument anonyme. En cas de dépôt en open-data, un nettoyage du fichier peut être réalisé, par exemple en ôtant les quasi-identifiants si ceux-ci n’ont servi qu’à produire des statistiques globales sur l’échantillon (p. ex. ratio H/F/autre). D’autres techniques de bluring sont décrites dans la référence donnée en ressource ci-dessous.
Pour aller plus loin…
Ce guideline s’inspire de l’article de Morehouse et al. (2025). Dans cet article de nombreux conseils sont donnés pour estimer et diminuer le risque de réidentification :
° Morehouse, K. N., Kurdi, B., & Nosek, B. A. (2025). Responsible data sharing: Identifying and remedying possible re-identification of human participants. American Psychologist, 80(6), 928–941. https://doi.org/10.1037/amp0001346
En 2008 Narayanan et Shmatikov alertaient sur les possibilités de réidentification, notamment dans les fichiers contenant beaucoup de quasi-identifiants. En utilisant l’Internet Movie Database comme source de connaissances de base, ils ont réussi à identifier les dossiers Netflix d’utilisateurs connus, révélant leurs préférences politiques apparentes ainsi que d’autres informations potentiellement sensibles.
° Narayanan, A., & Shmatikov, V. (2008). Robust De-anonymization of Large Sparse Datasets. 2008 IEEE Symposium on Security and Privacy (Sp 2008), 111–125. https://doi.org/10.1109/SP.2008.33
° Narayanan, A., & Shmatikov, V. (2019). Robust de-anonymization of large sparse datasets: A decade later. de-anonymization-retrospective.pdf
Pour aller plus loin sur le concept de k-anonymity :
° Sweeney, L. (2002). k-ANONYMITY: A MODEL FOR PROTECTING PRIVACY. International Journal of Uncertainty, Fuzziness and Knowledge-Based Systems, 10(05), 557–570. https://doi.org/10.1142/S0218488502001648
28. À quoi faut-il faire attention lors de l’utilisation de méthodes audiovisuelles en recherche ?
Du moment où sont produites des images sur lesquelles figurent des personnes reconnaissables, le responsable du projet de recherche doit respecter tant les principes de l’éthique de la recherche que les dispositions légales et réglementaires régissant la protection de la personnalité et le « droit à l’image » :
- Législation sur la protection des données personnelles : Loi fédérale sur la protection des données (LPD) au niveau national, Loi genevoise sur l’information du public l’accès aux documents et la protection des données personnelles (LIPAD) au niveau cantonal. L’image et la voix sont des données personnelles.
- Code civil suisse, art. 28 et suivants sur la protection de la personnalité.
Un troisième type de législation est à prendre en compte : il s’agit de la loi fédérale sur le droit d’auteur et les droits voisins (LDA), qui régit les questions liées au droit d’auteur. Ce point est abordé en fin de cette FAQ
Dans une recherche utilisant des méthodes (audio)visuelles, il s’agit donc de respecter à la fois les dispositions légales et les principes éthiques encadrant la collecte du consentement libre et éclairé/informé des participant-es à la recherche (ci-après, le consentement), et les conditions légales posées pour obtenir une autorisation valable de diffusion ou d’exploitation de l’image ou des images produites (ci-après, l’autorisation).
Tout comme le consentement, l’autorisation doit être donnée de manière libre et éclairée/informée. En principe, l’autorisation peut être également donnée par oral (par exemple, au début d’une séquence filmée), mais l’autorisation écrite est recommandée. L’autorisation doit au minimum comporter : le titre du projet et un court résumé du projet (« la recherche porte sur… »), le nom du chercheur/de la chercheuse responsable et l’institution dans le cadre de laquelle la recherche est menée (Université de Genève), quel type d’images sont prises (photographies, images vidéo), l’usage prévu des images (recherche uniquement/publications et diffusion en contexte académique/publications et diffusion grand public) et la durée d’utilisation (limitée ou illimitée dans le temps). Le-la participant-e peut demander des restrictions quant à l’usage tout en acceptant d’être photographié ou filmé.
S’agissant de personnes mineures ou sous curatelle, l’autorisation doit être signée par le représentant légal.
- Retrait de l’autorisation : une personne peut révoquer son autorisation en tout temps. Si la révocation intervient tardivement et cause un préjudice, par exemple financier, à la personne qui a réalisé ou diffusé les images, un juge peut être saisi. Dans ce cas, il examine la situation en mettant en balance les intérêts des deux parties : d’un côté, le préjudice ressenti par l’auteur des images et, de l’autre, celui invoqué par la personne qui y figure En cas, d’une utilisation d’images sans consentement la personne concernée peut se plaindre d’une atteinte illicite à sa personnalité (photos prises à l’insu de la personne, photos dites « de rue », etc.
S’agissant de photographies prises dans l’espace public : « si elles sont prises au su de toutes les personnes présentes et si les personnes représentées sur les photos n’en constituent pas le sujet principal (par ex., s’il s’agit de simples passants à proximité d’une curiosité locale), il est suffisant de supprimer la photo à la demande des personnes photographiées (immédiatement ou plus tard) ou de renoncer à sa publication. Il n’y a pas lieu cependant de les aborder exprès pour les informer de leurs droits » (https://www.edoeb.admin.ch/edoeb/fr/home/datenschutz/internet_technologie/umgang-fotos.html).
S’agissant de prises de vues ethnographiques effectuées dans un espace où circule un grand nombre de personnes, et où les demandes d’autorisation individuelles sont difficiles à demander (gare, lieux publics récréatifs, hôpital, etc.), le recours à une anonymisation à la prise de vue (contre-jour, flou de bougé, corps photographié partiellement) peut être une solution. Dans ce cas, la collaboration avec un-e professionnel-le de l’image est un atout. Toutefois, ces techniques d’anonymisation n’exemptent pas de l’obligation d’informer des prises de vues, par exemple par voie d’affichage dans le lieu en question, afin que les personnes ne souhaitant pas être photographiées ou filmées puissent se soustraire à l’objectif de l’appareil photo ou la caméra.
- Pour des raisons tant éthiques que légales, il est recommandé d’utiliser du « vrai » matériel photographique ou filmique pour prendre les images. Le smartphone n’est pas recommandé pour des raisons de protection des données (synchronisation avec un cloud et/ou d’autres appareils). De plus, prendre au sérieux les méthodes (audio)visuelles, c’est s’équiper avec du matériel adéquat, par exemple dans le cadre d’un budget FNS ou autre.
Le problème est-il réglé si j’anonymise tout en floutant les images photographiques ou filmiques ?
Non, il faut quand même demander l’autorisation de photographier/filmer et, dans le formulaire d’autorisation, préciser que la personne sera rendue méconnaissable par floutage. S’il s’agit de vidéo, il convient de préciser si la voix sera transformée aussi pour assurer l’anonymat.
- Flouter des personnes sans les informer peut aussi être considéré comme une atteinte à la personnalité et au droit à l’image (la personne peut estimer être lésée).
Dans tous les cas, il convient de prendre le temps de réfléchir à la place des images dans le projet de recherche, au choix à faire entre images « à visage découvert » et images anonymisées, à l’usage des images au cours du projet et durant la phase de valorisation du projet, choix qui doivent être exposés et négociés sur le terrain. L’anonymisation par floutage ne doit pas être une facilité légale mais un vrai choix.
Comment faire si je veux utiliser des méthodes (audio)visuelles dans le cadre d’une démarche de recherche participative ?
Si la réalisation des images photographiques ou filmiques est confiée à des participant-es à la recherche, il convient d’être très attentif à la délégation de l’obligation de recueillir le consentement et l’autorisation nécessaires. Si les participant-es se photographient/filment eux-mêmes (auto-ethnographie), le consentement et l’autorisation des participant-es suffisent. Mais du moment où ils et elles prennent des images de tierces personnes, même brièvement ou fortuitement, même si ce sont des proches (famille, ami-es), il convient de recueillir les consentements et les autorisations nécessaires auprès de ces tierces personnes. Les participant-es doivent être soigneusement formés et informés car, ils et elles deviennent légalement responsables des images qu’ils et elles prennent.
Le chercheur ou la chercheuse utilisant des méthodes (audio)visuelles dans le cadre d’une démarche de recherche participative devrait avoir a) une bonne connaissance de son terrain de recherche, b) une bonne connaissance des méthodes visuelles et c) une bonne connaissance du cadre légal audiovisuel.
Qui détient les droits sur les images réalisées dans le cadre d’une recherche (audio)visuelle ?
Les images photographiques ou filmiques sont associées à des œuvres et protégées par la loi fédérale sur le droit d’auteur et les droits voisins. Le titulaire des droits est l’auteur-l’autrice des images et les droits d’auteur moraux (droit à la reconnaissance de la paternité intellectuelle de l’œuvre, droit de décider de la divulgation d’une œuvre et droit au respect de l’intégrité de l’œuvre) sont perpétuels et inaliénables, autrement dit, ils ne peuvent pas être cédés à un tiers. En revanche, les droits d’auteur patrimoniaux (droit d’utilisation, de diffusion, d’exploitation, etc.) peut être cédés (par exemple, à une revue pour publication).
Dans le cas de méthodes participatives, les participant-es ayant fait des images détiennent les droits d’auteur sur ces images. Il convient donc de leur demander de céder les droits d’utilisation, de reproduction et de diffusion des images au chercheur/à la chercheuse par le biais d’un formulaire de cession de droits.
A quoi dois-je prêter attention lorsque je traite, stocke et archive des données visuelles ?
Si des personnes figurent sur les images photographiques ou filmiques, ces images doivent être traitées et stockées de façon sécurisée, puisqu’elles constituent des données personnelles et/ou sensibles. Le niveau de sécurité et de confidentialité des logiciels de postproduction (traitement de l’image, montage audiovisuel) est à contrôler.
S’agissant de l’archivage, la durée de l’archivage des images doit correspondre à ce qui figure sur l’autorisation mentionnée plus haut. La durée peut donc dépasser les 12 mois de conservation des données mentionnés dans le formulaire CUREG. On recommandera toutefois de discuter cette durée avec les participant-es, en ayant à l’esprit que la notion de « droit à l’oubli » commence à être un thème important dans le domaine de l’audiovisuel. Il convient donc faire une pesée d’intérêts entre la valeur possiblement historique (histoire scientifique de la discipline) des images et les intérêts privés des participant-es. Il est également possible d’envisager deux durées d’archivages différentes selon le statut des données : archivage court pour l’ensemble des données brutes (toutes les photos, toutes les séquences filmiques nommées « rushes » dans le jargon) et archivage long pour un « output » spécifique de la recherche : livre, exposition, film documentaire.
Remarque finale :
Cette FAQ reste à un haut niveau de généralité. Selon les disciplines et les domaines de recherche, des procédures précises peuvent avoir été développées (par exemple, les recherches en contexte scolaire en sciences de l’éducation, les recherches dans le domaine des médias en sciences sociales). Les chercheurs et chercheuses sont invité-es à se renseigner sur l’existence de procédures ou bonnes pratiques dans leur domaine de recherche lors de la rédaction du projet de recherche.